Accreditation for Microbiological Laboratories (2023)

Contents
This Eurachem Guide has been produced by an ad hoc Eurachem Working Group. This third edition of Accreditation for Microbiological Laboratories is a revision of the second edition published in 2013. The first edition from 2002 was produced by a joint EA/Eurachem Working Group.
The aim of this guide is to provide microbiological laboratories with guidance on how to fulfil the requirements of ISO/IEC 17025. The content, however, should also be of use to organisations seeking accreditation or certification against the requirements of standards such as ISO 15189, GLP (Good Laboratory Practice), GMP (Good Manufacturing Practice), and GCP (Good Clinical Practice)). Specific national regulations may override the guidance given in this document. The Guide will also provide useful information for laboratories that wish to establish a quality management system but are not seeking formal recognition.
The principal changes in the third edition are:
- update on recent trends in microbiology, e.g. PCR (polymerase chain reaction) techniques for the detection of microorganisms;
- addition of a list of abbreviations and symbols;
- addition of a section on risk-based thinking;
- updated sections on method verification and validation to reflect current ISO standards;
- references to the use of a decision rule;
- updated Annex A on terminology relevant to microbiology;
- new Annex C on reporting confidence intervals;
- new Annex D on estimation of uncertainty from sampling;
- the order of sections in adherence with ISO/IEC 17025:2017.
Availability
This third edition is currently available in English and can be downloaded at no cost from the link below:
Download the guide in English [pdf, 272 kB] (2023-05-23)+
+ Date of publication on this site
Translations
Translation into other languages is permitted for members of Eurachem. Other offers of translation should be directed to the Eurachem Secretariat for permission. The Eurachem policy on maintenance and development of Eurachem guidance, available on the Policies page, gives further information on translation.
Citation
This guidance should be cited* as:
“B Magnusson and K C Tsimillis (eds.) Accreditation for Microbiological Laboratories (3rd ed. 2023). ISBN 978-91-519-6581-9. Available from www.eurachem.org.”
*Subject to journal requirements
Previous versions
The second edition of this Guide can be found in the publication archive on this website.


 Many decisions with socio-economic or individual impact depend on qualitative analysis, including decisions related to food safety, clinical diagnosis, and forensic evidence, are based primarily on qualitative, rather than quantitative, chemical analysis. Qualitative analysis is analysis that returns a classification rather than a numerical value, such as the identity of a chemical substance, the type of plastic of a microparticle, the potential source of an oil spill, the presence of a banned sports doping substance, or the presence of accelerant in fire debris. Sometimes, such classifications rely solely on qualitative tests; others may use measurement results (such as line frequencies in a spectrum) to reach a conclusion.
Many decisions with socio-economic or individual impact depend on qualitative analysis, including decisions related to food safety, clinical diagnosis, and forensic evidence, are based primarily on qualitative, rather than quantitative, chemical analysis. Qualitative analysis is analysis that returns a classification rather than a numerical value, such as the identity of a chemical substance, the type of plastic of a microparticle, the potential source of an oil spill, the presence of a banned sports doping substance, or the presence of accelerant in fire debris. Sometimes, such classifications rely solely on qualitative tests; others may use measurement results (such as line frequencies in a spectrum) to reach a conclusion.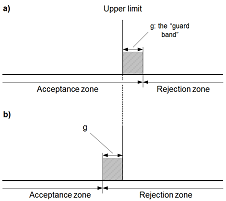 In order to decide whether a result indicates compliance or non-compliance with a specification, it is necessary to take into account the measurement uncertainty associated with the result. This guide provides guidance on how uncertainty may be taken into account in deciding compliance with a limit.
In order to decide whether a result indicates compliance or non-compliance with a specification, it is necessary to take into account the measurement uncertainty associated with the result. This guide provides guidance on how uncertainty may be taken into account in deciding compliance with a limit. Blanks are an important tool and are used in the determination of most performance characteristics during a validation process. They are also often included in each analytical run during routine use of the measurement procedure. There are many different types of blanks and the analyst must consider which blanks to include during preparation of the validation plan.
Blanks are an important tool and are used in the determination of most performance characteristics during a validation process. They are also often included in each analytical run during routine use of the measurement procedure. There are many different types of blanks and the analyst must consider which blanks to include during preparation of the validation plan.